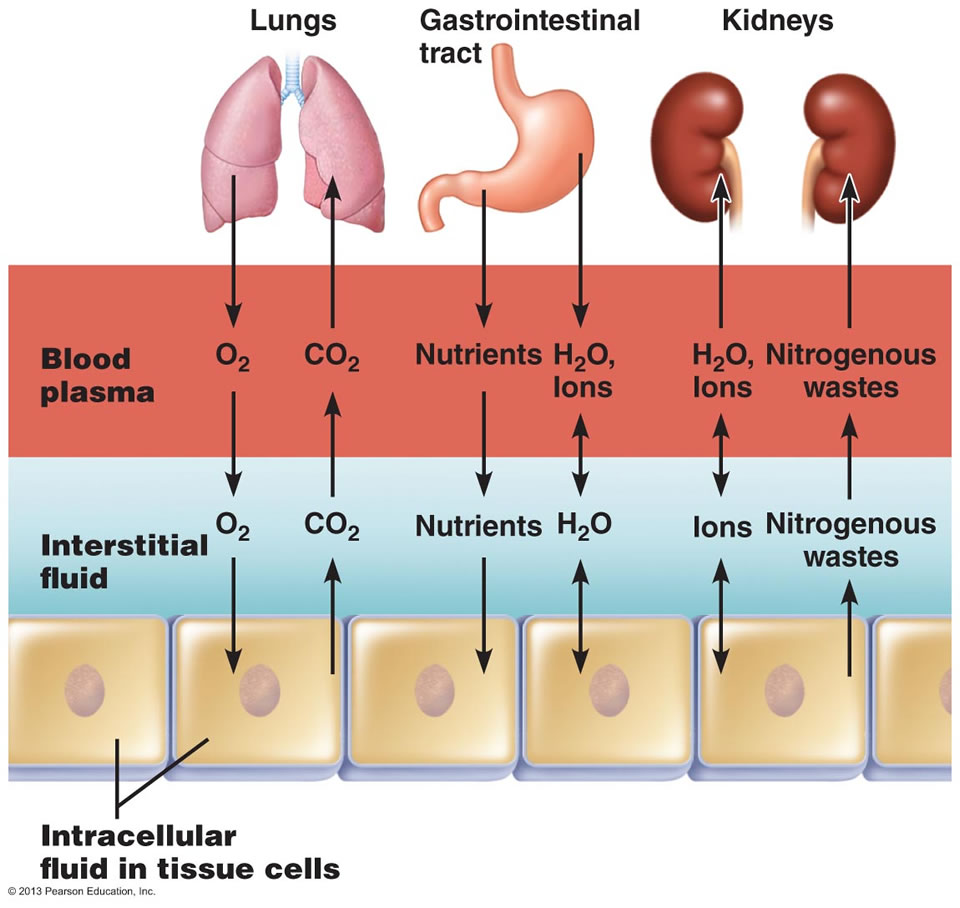 
The membranes are highly permeable to water but require energy to transport ions. Fluid is separated into compartments by semipermeable membranes.On the right is an image representing the exchange of water among body fluid compartments. On the left is an image showing the percentage of distribution of body fluids in males and females, in different compartments. Principal component of the cytoplasm of cellsīody fluid compartments Image by Lecturio.Interstitial fluid (¾ of ECF): lies outside blood vessels.Intravascular fluid (¼ of ECF): primary component of plasma Plasma The residual portion of blood that is left after removal of blood cells by centrifugation without prior blood coagulation.The total body water is distributed primarily between 2 compartments, namely, extracellular fluid (ECF) and intracellular fluid (ICF) compartments. Hypernatremia of the body fluid compartments. Osmolality is expressed in terms of osmoles of solute per kilogram of solvent. Edema are the clinical conditions arising from disturbances in the maintenance of osmolarity Osmolarity The concentration of osmotically active particles in solution expressed in terms of osmoles of solute per liter of solution. It can be categorized into 2 types, namely, peripheral (in the extremities) and internal (in an organ or body cavity). Edema is a symptom observed in several medical conditions. Hyponatremia, and edema Edema Edema is a condition in which excess serous fluid accumulates in the body cavity or interstitial space of connective tissues. Serum sodium is the greatest contributor to plasma osmolality, which is very tightly controlled via antidiuretic hormone (ADH) release from the hypothalamus and by the thirst mechanism. Hypernatremia, hyponatremia Hyponatremia Hyponatremia is defined as a decreased serum sodium (sNa ) concentration less than 135 mmol/L. Hypernatremia occurs either from a lack of access to water or an excessive intake of sodium. Serum sodium is the greatest contributor to plasma osmolality, which is very tightly controlled by the hypothalamus via the thirst mechanism and antidiuretic hormone (ADH) release. Hypernatremia Hypernatremia Hypernatremia is an elevated serum sodium concentration > 145 mmol/L. Hypernatremia are maintained rigorously by the transport of water and substances between compartments. The relative charge and osmolarity Osmolarity The concentration of osmotically active particles in solution expressed in terms of osmoles of solute per liter of solution. Each compartment contains different concentrations of ions and osmolar molecules. A cell membrane is composed of a phospholipid bilayer and proteins that function to protect cellular DNA and mediate the exchange of ions and molecules. Intracellular and extracellular fluids are separated into compartments by semipermeable membranes, and the transport of fluid and ions is maintained by channels Channels The Cell: Cell Membrane in the cell membrane Cell Membrane A cell membrane (also known as the plasma membrane or plasmalemma) is a biological membrane that separates the cell contents from the outside environment. Intracellular fluid is present inside the cells and makes up ⅔ of the total body water. Extracellular fluid is present outside the cells and makes up ⅓ of the total body water. The adult human body is made up of 60% water and is divided into extracellular and intracellular fluid compartments. Students: Educators’ Pro Tips for Tough Topics.Maternity Nursing and Care of the Childbearing Family.What should the voltage at the center of the accelerator be, assuming that the ions start essentially at rest? (a) -3.0 MV (b) 3.0 MV (c) 1.5 MV (d) 1.0 MV. For a particular experiment, helium ions are to be given a kinetic energy of 3.0 MeV. These positive ions are then repelled from the midpoint by the high positive voltage there and continue to accelerate to the far end of the accelerator, where again V = 0. In this device, negative ions ( H e − ) \left(\mathrm\right) ( He ). To accelerate the ions to high energies, a tandem electrostatic accelerator may be used. By analyzing the distribution and energy of the ions that are scattered from (that is, deflected by collisions with) the atoms in the sample, researchers can determine the sample’s composition. A beam of ions (typically helium ions) is accelerated to high energy and aimed at a sample. Rutherford backscattering spectrometry (RBS) is a technique used to determine the structure and composition of materials.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
